New Gut Health Insights from Groundbreaking Research
Hettich predicts that within five years, doctors may routinely sequence patients’ microbiomes using faecal samples. This would provide insights into health conditions such as cancer, diabetes, and autoimmune diseases.

The Human Genome and Microbiome:
A Powerful Partnership
The human genome contains approximately 23,000 genes—only slightly more than simpler organisms. However, our bodies host around 1 million additional genes contributed by bacteria and other microorganisms in our digestive system. These microbial genes significantly expand our functional abilities, enabling us to digest foods like salads and other plant-based polysaccharides without discomfort.

This fascinating interplay between human and microbial genes was the focus of a recent talk by Bob Hettich, a corporate fellow and leader of the Bioanalytical Mass Spectrometry Group at Oak Ridge National Laboratory (ORNL). Supported by the National Institutes of Health, Hettich’s research explores the human gut microbiome using advanced biological mass spectrometry techniques.
Gut Microbiome:
A Window into Health
Hettich predicts that within the next five years, doctors may routinely sequence patients’ microbiomes using feacal samples. This analysis would reveal the types and populations of bacterial proteins in the gut, providing valuable insights into health conditions such as cancer, diabetes, and autoimmune diseases like Crohn’s or Parkinson’s.
“Healthy individuals have a rich diversity of bacteria in their gut, predominantly in the colon,” Hettich explained. In contrast, people with conditions like diabetes or Crohn’s disease experience a significant reduction in bacterial diversity, leading to a less complex microbiome. A thinner mucous layer in the colon exacerbates inflammation diseases such as Crohn’s or ulcerative colitis.
By understanding the bacterial composition of a patient’s microbiome, doctors can better tailor treatments, determining which medications will be effective and which will not.
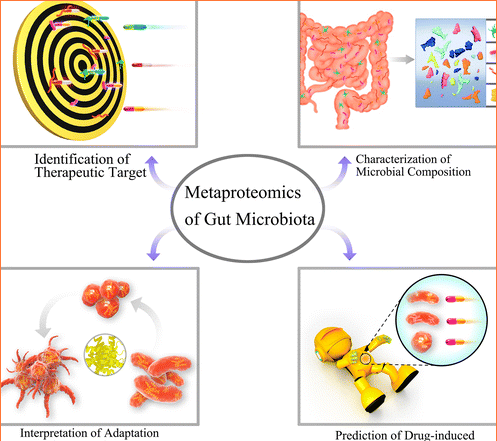
Metaproteomics:
A Revolutionary approach
Hettich is a pioneer in metaproteomics—a method that uses biological mass spectrometry to identify and quantify proteins in complex biological samples like the gut microbiome. This approach helps researchers understand how microorganisms function within their communities.
A Personal Connection to the Research
Hettich's passion for this work stems from personal experience—his daughter was diagnosed with Crohn's disease at just eight years old, unusually early for this condition that typically appears in the late teens to early twenties. She required multiple surgeries and treatments.
He believes her condition may have originated from a bacterial infection during birth that led to a life-threatening blood infection. After an initial antibiotic proved ineffective, doctors administered a stronger one that saved her life but potentially disrupted her developing microbiome.
Working with pediatric surgeon Michael Morowitz from the University of Pittsburgh, Hettich has studied microbiome development in infants. Their research suggests that overusing antibiotics in babies and young children—such as for frequent ear infections—can negatively impact microbiome development and long-term health.
Hettich noted that Southeastern U.S. residents have poor microbiome health due to high rates of antibiotic use and diets high in sugar and fat, which reduce beneficial bacterial diversity. These factors contribute to various health conditions, including obesity.
The Microbiome's Early Development
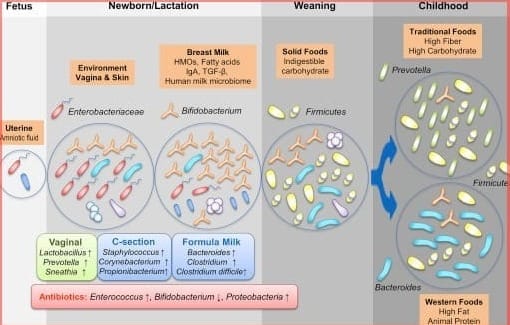
Collaborating with Dr. Michael Morowitz, a pediatric surgeon at the University of Pittsburgh, Hettich has studied how microbiomes evolve in infants—particularly preterm babies. Surprisingly, most babies are born with sterile guts despite the abundance of bacteria in their mothers’ wombs. Within a week of birth, however, their guts are colonized by bacteria from environmental sources such as caregivers, feeding methods, and even pacifiers.
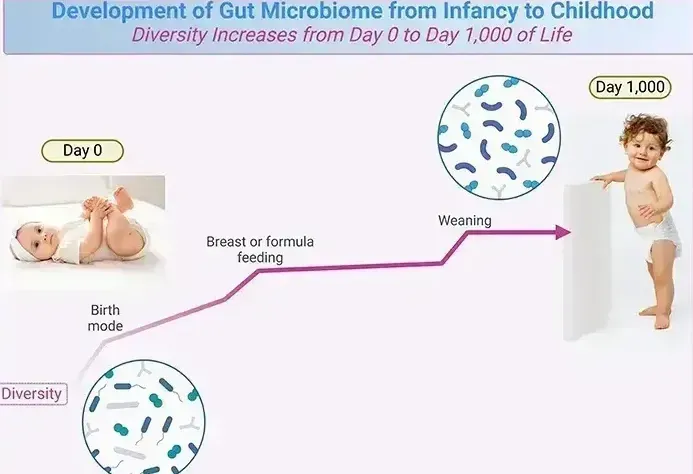
The mode of delivery also plays a role: vaginally delivered babies acquire microbiomes resembling their mother’s faecal bacteria, while those born via Caesarean section develop microbiomes similar to their mother’s skin bacteria. Breastfeeding versus formula feeding further influences microbiome composition.
One alarming condition affecting preterm infants is necrotizing enterocolitis (NEC), a severe gastrointestinal disease that can lead to intestinal tissue death and is often fatal. In one study involving 16 preterm infants, Hettich’s team used metaproteomics to track changes in their microbiomes over three months. They discovered that even identical twins could have vastly different gut bacteria compositions—one twin might survive an infection while the other succumbs to NEC.
Antibiotics and Microbiome Health
The overuse of antibiotics in infants and young children can disrupt the delicate balance of their developing microbiomes, potentially leading to long-term health issues such as obesity or inflammatory bowel diseases. Residents of certain regions are more prone to these disruptions due to high antibiotic use and diets rich in sugar and fat—factors that diminish microbial diversity essential for good health.

The Role of Bacteria in Gut Health
Hettich highlighted how beneficial bacteria like Bacteroides species act as “500-pound gorillas” that dominate the gut environment and keep harmful bacteria such as Clostridium difficile at bay. Interestingly, his team found that white blood cells use copper to selectively kill certain bacteria during early microbiome development, allowing beneficial microbes to thrive—a potential natural mechanism for maintaining microbial balance even in preterm infants.
Why Microorganisms Matter
While some may find it unsettling to learn that microbial cells outnumber human cells by three to ten times in our bodies, these microorganisms play vital roles in digestion, infection prevention, and overall health. As research progresses, understanding our microbiomes could revolutionize medicine and offer new hope for managing chronic diseases.
This article provides an engaging yet professional summary of Bob Hettich's groundbreaking research on gut health and its implications for modern medicine.